what happens to the electrons as they move down the electron transport chain

An electron transport chain (ETC [1]) is a series of protein complexes and other molecules that transfer electrons from electron donors to electron acceptors via redox reactions (both reduction and oxidation occurring simultaneously) and couples this electron transfer with the transfer of protons (H+ ions) beyond a membrane. Many of the enzymes in the electron send chain are membrane-bound.
The flow of electrons through the electron transport chain is an exergonic process. The energy from the redox reactions creates an electrochemical proton gradient that drives the synthesis of adenosine triphosphate (ATP). In aerobic respiration, the flow of electrons terminates with molecular oxygen as the concluding electron acceptor that provides most of the energy.[2] In anaerobic respiration, other, lower-energy electron acceptors are used, such every bit sulfate.
In an electron send chain, the redox reactions are driven past the deviation in the Gibbs free free energy of reactants and products. The complimentary energy released when a college-energy electron donor and acceptor catechumen to lower-energy products, while electrons are transferred from a lower to a college redox potential, is used by the complexes in the electron transport chain to create an electrochemical gradient of ions. Information technology is this electrochemical gradient that drives the synthesis of ATP via coupling with oxidative phosphorylation with ATP synthase.[3]
In eukaryotic organisms the electron transport chain, and site of oxidative phosphorylation, is establish on the inner mitochondrial membrane. The energy of oxygen released in its reaction with reduced compounds such equally cytochrome c and (indirectly) NADH and FADH is used past the electron ship chain to pump protons into the intermembrane space, generating the electrochemical gradient over the inner mitochondrial membrane. In photosynthetic eukaryotes, the electron transport chain is plant on the thylakoid membrane. Here, light energy drives electron send through a proton pump and the resulting proton gradient causes subsequent synthesis of ATP. In bacteria, the electron transport chain can vary betwixt species but it ever constitutes a set of redox reactions that are coupled to the synthesis of ATP through the generation of an electrochemical gradient and oxidative phosphorylation through ATP synthase.[iv]
Mitochondrial electron transport chains [edit]
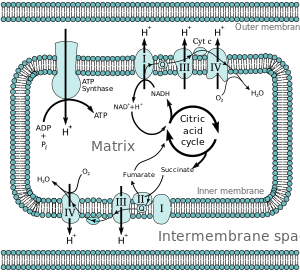
Nearly eukaryotic cells have mitochondria, which produce ATP from reactions of oxygen with products of the citric acrid cycle, fatty acid metabolism, and amino acid metabolism. At the inner mitochondrial membrane, electrons from NADH and FADH2 laissez passer through the electron transport chain to oxygen, which provides the free energy driving the process as it is reduced to water.[5] The electron transport concatenation comprises an enzymatic series of electron donors and acceptors. Each electron donor will laissez passer electrons to an acceptor of college redox potential, which in turn donates these electrons to another acceptor, a procedure that continues down the series until electrons are passed to oxygen, the most energy-rich[2] and final electron acceptor in the concatenation. Each reaction releases energy considering a higher-energy donor and acceptor convert to lower-free energy products. Via the transferred electrons, this free energy is used to generate a proton gradient across the mitochondrial membrane past "pumping" protons into the intermembrane space, producing a country of higher free energy that has the potential to do piece of work. This unabridged process is called oxidative phosphorylation since ADP is phosphorylated to ATP by using the electrochemical gradient that the redox reactions of the electron transport chain have established driven past the free energy of oxygen.
Mitochondrial redox carriers [edit]
Free energy associated with the transfer of electrons downwardly the electron send chain is used to pump protons from the mitochondrial matrix into the intermembrane space, creating an electrochemical proton gradient (ΔpH) beyond the inner mitochondrial membrane. This proton gradient is largely but not exclusively responsible for the mitochondrial membrane potential (ΔΨM).[half-dozen] It allows ATP synthase to use the flow of H+ through the enzyme back into the matrix to generate ATP from adenosine diphosphate (ADP) and inorganic phosphate. Circuitous I (NADH coenzyme Q reductase; labeled I) accepts electrons from the Krebs cycle electron carrier nicotinamide adenine dinucleotide (NADH), and passes them to coenzyme Q (ubiquinone; labeled Q), which also receives electrons from Circuitous II (succinate dehydrogenase; labeled Ii). Q passes electrons to Complex III (cytochrome bc1 complex; labeled 3), which passes them to cytochrome c (cyt c). Cyt c passes electrons to Complex 4 (cytochrome c oxidase; labeled 4), which uses the electrons and hydrogen ions to release the energy of molecular oxygen equally it is reduced to h2o.
Four membrane-bound complexes take been identified in mitochondria. Each is an extremely complex transmembrane structure that is embedded in the inner membrane. Three of them are proton pumps. The structures are electrically connected by lipid-soluble electron carriers and water-soluble electron carriers. The overall electron transport concatenation can be summarized equally follows:
NADH+H+ → Complex I → Q ↑ Complex 2 ↑ Succinate → Complex Three → cytochrome c → Complex 4 → H2O ↑ Complex II ↑ Succinate
Complex I [edit]
In Complex I (NADH ubiquinone oxidoreductase, Type I NADH dehydrogenase, or mitochondrial complex I; EC 1.6.5.3), two electrons are removed from NADH and transferred to a lipid-soluble carrier, ubiquinone (Q). The reduced product, ubiquinol (QH2), freely diffuses within the membrane, and Circuitous I translocates iv protons (H+) beyond the membrane, thus producing a proton slope. Circuitous I is one of the chief sites at which premature electron leakage to oxygen occurs, thus being 1 of the main sites of product of superoxide.[7]
The pathway of electrons is as follows:
NADH is oxidized to NAD+, past reducing flavin mononucleotide to FMNH2 in one 2-electron step. FMNHtwo is then oxidized in ii one-electron steps, through a semiquinone intermediate. Each electron thus transfers from the FMNHii to an Fe–S cluster, from the Fe-South cluster to ubiquinone (Q). Transfer of the first electron results in the complimentary-radical (semiquinone) form of Q, and transfer of the second electron reduces the semiquinone form to the ubiquinol form, QH2. During this process, four protons are translocated from the mitochondrial matrix to the intermembrane space.[eight] As the electrons move through the complex an electron current is produced along the 180 Angstrom width of the complex inside the membrane. This electric current powers the active transport of four protons to the intermembrane infinite per two electrons from NADH.[ix]
Circuitous Ii [edit]
In Complex Ii (succinate dehydrogenase or succinate-CoQ reductase; EC 1.three.5.1) additional electrons are delivered into the quinone pool (Q) originating from succinate and transferred (via flavin adenine dinucleotide (FAD)) to Q. Circuitous Two consists of iv protein subunits: succinate dehydrogenase, (SDHA); succinate dehydrogenase [ubiquinone] atomic number 26–sulfur subunit, mitochondrial, (SDHB); succinate dehydrogenase complex subunit C, (SDHC) and succinate dehydrogenase complex, subunit D, (SDHD). Other electron donors (e.g., fatty acids and glycerol 3-phosphate) also direct electrons into Q (via FAD). Complex 2 is a parallel electron ship pathway to complex i, simply dissimilar Complex I, no protons are transported to the intermembrane space in this pathway. Therefore, the pathway through Complex II contributes less energy to the overall electron transport chain procedure.
Complex 3 [edit]
In Complex III (cytochrome bcone complex or CoQHtwo-cytochrome c reductase; EC 1.ten.2.2), the Q-bicycle contributes to the proton gradient by an asymmetric assimilation/release of protons. Two electrons are removed from QH2 at the QO site and sequentially transferred to ii molecules of cytochrome c, a water-soluble electron carrier located inside the intermembrane space. The two other electrons sequentially pass across the protein to the Qi site where the quinone part of ubiquinone is reduced to quinol. A proton gradient is formed past ane quinol ( ) oxidations at the Qo site to form one quinone ( ) at the Qi site. (In total, four protons are translocated: two protons reduce quinone to quinol and two protons are released from two ubiquinol molecules.)
When electron transfer is reduced (by a high membrane potential or respiratory inhibitors such as antimycin A), Complex Three may leak electrons to molecular oxygen, resulting in superoxide germination.
This complex is inhibited by dimercaprol (British Antilewisite, BAL), Napthoquinone and Antimycin.
Complex IV [edit]
In Complex IV (cytochrome c oxidase; EC one.9.three.1), sometimes called cytochrome AA3, four electrons are removed from iv molecules of cytochrome c and transferred to molecular oxygen (Oii) and 4 protons, producing 2 molecules of water. The complex contains coordinated copper ions and several heme groups. At the same time, the energy of Oii is used to remove eight protons from the mitochondrial matrix (although only four are translocated across the membrane), contributing to the proton gradient. The verbal details of proton pumping in Complex IV are still under report.[10] Cyanide is an inhibitor of Circuitous IV.
Coupling with oxidative phosphorylation [edit]
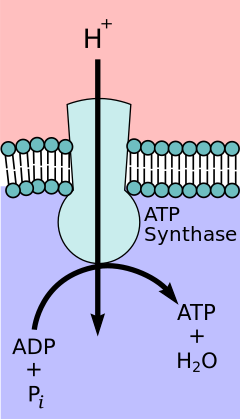
Depiction of ATP synthase, the site of oxidative phosphorylation to generate ATP.
According to the chemiosmotic coupling hypothesis, proposed by Nobel Prize in Chemistry winner Peter D. Mitchell, the electron transport concatenation and oxidative phosphorylation are coupled past a proton gradient across the inner mitochondrial membrane. The efflux of protons from the mitochondrial matrix creates an electrochemical gradient (proton slope). This gradient is used past the FOFane ATP synthase circuitous to brand ATP via oxidative phosphorylation. ATP synthase is sometimes described equally Complex V of the electron transport chain.[eleven] The FO component of ATP synthase acts equally an ion channel that provides for a proton flux back into the mitochondrial matrix. It is composed of a, b and c subunits. Protons in the inter-membrane space of mitochondria showtime enter the ATP synthase complex through an a subunit channel. Then protons move to the c subunits.[12] The number of c subunits determines how many protons are required to make the FO turn one full revolution. For case, in humans, in that location are 8 c subunits, thus eight protons are required.[13] After c subunits, protons finally enter the matrix through an a subunit channel that opens into the mitochondrial matrix.[12] This reflux releases costless energy produced during the generation of the oxidized forms of the electron carriers (NAD+ and Q) with free energy provided by O2. The free free energy is used to drive ATP synthesis, catalyzed by the Fane component of the complex.[14]
Coupling with oxidative phosphorylation is a cardinal step for ATP production. However, in specific cases, uncoupling the ii processes may be biologically useful. The uncoupling protein, thermogenin—nowadays in the inner mitochondrial membrane of chocolate-brown adipose tissue—provides for an alternative menstruum of protons back to the inner mitochondrial matrix. Thyroxine is also a natural uncoupler. This alternative catamenia results in thermogenesis rather than ATP production.[xv]
Opposite electron flow [edit]
Reverse electron flow is the transfer of electrons through the electron transport chain through the reverse redox reactions. Usually requiring a pregnant amount of energy to be used, this can reduce the oxidized forms of electron donors. For example, NAD+ can be reduced to NADH past Circuitous I.[16] In that location are several factors that have been shown to induce opposite electron flow. However, more piece of work needs to be washed to ostend this. One case is blockage of ATP synthase, resulting in a build-upwardly of protons and therefore a higher proton-motive force, inducing opposite electron catamenia.[17]
Bacterial electron transport chains [edit]
In eukaryotes, NADH is the most important electron donor. The associated electron transport concatenation is NADH → Complex I → Q → Complex III → cytochrome c → Complex IV → O2 where Complexes I, III and IV are proton pumps, while Q and cytochrome c are mobile electron carriers. The electron acceptor providing the energy for this process is molecular oxygen.
In prokaryotes (bacteria and archaea) the state of affairs is more complicated, because in that location are several different electron donors and several different electron acceptors. The generalized electron transport chain in leaner is:
Donor Donor Donor ↓ ↓ ↓ dehydrogenase → quinone → bc1 → cytochrome ↓ ↓ oxidase(reductase) oxidase(reductase) ↓ ↓ Acceptor Acceptor
Electrons tin enter the chain at iii levels: at the level of a dehydrogenase, at the level of the quinone pool, or at the level of a mobile cytochrome electron carrier. These levels represent to successively more than positive redox potentials, or to successively decreased potential differences relative to the concluding electron acceptor. In other words, they correspond to successively smaller Gibbs free energy changes for the overall redox reaction.
Individual bacteria apply multiple electron transport bondage, ofttimes simultaneously. Bacteria can use a number of different electron donors, a number of different dehydrogenases, a number of different oxidases and reductases, and a number of different electron acceptors. For example, Due east. coli (when growing aerobically using glucose and oxygen as an energy source) uses two different NADH dehydrogenases and two different quinol oxidases, for a total of four dissimilar electron send chains operating simultaneously.
A common feature of all electron transport bondage is the presence of a proton pump to create an electrochemical gradient over a membrane. Bacterial electron transport chains may comprise as many as three proton pumps, like mitochondria, or they may incorporate two or at least one.
Electron donors [edit]
In the current biosphere, the most common electron donors are organic molecules. Organisms that apply organic molecules every bit an electron source are called organotrophs. Chemoorganotrophs (animals, fungi, protists) and photolithotrophs (plants and algae) plant the vast majority of all familiar life forms.
Some prokaryotes can use inorganic thing as an electron source. Such an organism is chosen a (chemo)lithotroph ("rock-eater"). Inorganic electron donors include hydrogen, carbon monoxide, ammonia, nitrite, sulfur, sulfide, manganese oxide, and ferrous fe. Lithotrophs accept been found growing in rock formations thousands of meters beneath the surface of Earth. Because of their book of distribution, lithotrophs may really outnumber organotrophs and phototrophs in our biosphere.
The employ of inorganic electron donors such as hydrogen as an energy source is of particular interest in the study of evolution. This type of metabolism must logically have preceded the use of organic molecules and oxygen every bit an energy source.
Complexes I and Ii [edit]
Leaner can use several different electron donors. When organic matter is the electron source, the donor may exist NADH or succinate, in which case electrons enter the electron transport chain via NADH dehydrogenase (similar to Complex I in mitochondria) or succinate dehydrogenase (similar to Complex II). Other dehydrogenases may be used to process different free energy sources: formate dehydrogenase, lactate dehydrogenase, glyceraldehyde-3-phosphate dehydrogenase, H2 dehydrogenase (hydrogenase), electron ship chain. Some dehydrogenases are also proton pumps, while others funnel electrons into the quinone puddle. Most dehydrogenases show induced expression in the bacterial cell in response to metabolic needs triggered by the surroundings in which the cells grow. In the instance of lactate dehydrogenase in E. coli, the enzyme is used aerobically and in combination with other dehydrogenases. It is inducible and is expressed when the concentration of DL-lactate in the jail cell is high.[ citation needed ]
Quinone carriers [edit]
Quinones are mobile, lipid-soluble carriers that shuttle electrons (and protons) between big, relatively immobile macromolecular complexes embedded in the membrane. Bacteria use ubiquinone (Coenzyme Q, the same quinone that mitochondria utilise) and related quinones such every bit menaquinone (Vitamin K<2). Archaea in the genus Sulfolobus utilise caldariellaquinone.[18] The apply of dissimilar quinones is due to slight changes in redox potentials caused past changes in construction. The change in redox potentials of these quinones may be suited to changes in the electron acceptors or variations of redox potentials in bacterial complexes.[xix]
Proton pumps [edit]
A proton pump is any process that creates a proton slope across a membrane. Protons can be physically moved across a membrane, as seen in mitochondrial Complexes I and IV. The aforementioned effect can be produced by moving electrons in the opposite direction. The result is the disappearance of a proton from the cytoplasm and the appearance of a proton in the periplasm. Mitochondrial Circuitous Three uses this 2d type of proton pump, which is mediated by a quinone (the Q bicycle).
Some dehydrogenases are proton pumps, while others are not. Most oxidases and reductases are proton pumps, but some are not. Cytochrome bc1 is a proton pump found in many, but not all, leaner (not in E. coli). As the proper name implies, bacterial bc1 is like to mitochondrial bc1 (Complex III).
Cytochrome electron carriers [edit]
Cytochromes are proteins that contain iron. They are plant in two very different environments.
Some cytochromes are water-soluble carriers that shuttle electrons to and from large, immobile macromolecular structures imbedded in the membrane. The mobile cytochrome electron carrier in mitochondria is cytochrome c. Bacteria use a number of dissimilar mobile cytochrome electron carriers.
Other cytochromes are found within macromolecules such equally Complex III and Circuitous 4. They also function equally electron carriers, but in a very different, intramolecular, solid-land environment.
Electrons may enter an electron ship chain at the level of a mobile cytochrome or quinone carrier. For example, electrons from inorganic electron donors (nitrite, ferrous atomic number 26, electron transport chain) enter the electron ship concatenation at the cytochrome level. When electrons enter at a redox level greater than NADH, the electron send concatenation must operate in reverse to produce this necessary, college-energy molecule.
Terminal oxidases and reductases [edit]
When bacteria abound in aerobic environments, the terminal electron acceptor (O2) is reduced to h2o by an enzyme called an oxidase. When bacteria grow in anaerobic environments, the final electron acceptor is reduced by an enzyme chosen a reductase. In mitochondria the final membrane circuitous (Circuitous IV) is cytochrome oxidase. Aerobic leaner use a number of dissimilar terminal oxidases. For example, E. coli (a facultative anaerobe) does not have a cytochrome oxidase or a bc1 complex. Under aerobic conditions, it uses two different terminal quinol oxidases (both proton pumps) to reduce oxygen to water.
Bacterial Complex IV can exist split up into classes according to the molecules human activity every bit terminal electron acceptors. Course I oxidases are cytochrome oxidases and utilize oxygen as the terminal electron acceptor. Class 2 oxidases are quinol oxidases and can use a diversity of terminal electron acceptors. Both of these classes tin be subdivided into categories based on what redox-agile components they contain. E.g. Heme aa3 Class i concluding oxidases are much more efficient than Class 2 concluding oxidases[3]
Anaerobic bacteria, which do not apply oxygen equally a terminal electron acceptor, accept terminal reductases individualized to their terminal acceptor. For example, E. coli tin use fumarate reductase, nitrate reductase, nitrite reductase, DMSO reductase, or trimethylamine-N-oxide reductase, depending on the availability of these acceptors in the environment.
Most terminal oxidases and reductases are inducible. They are synthesized by the organism as needed, in response to specific environmental atmospheric condition.
Electron acceptors [edit]
Just every bit in that location are a number of different electron donors (organic matter in organotrophs, inorganic matter in lithotrophs), there are a number of unlike electron acceptors, both organic and inorganic. If oxygen is available, it is invariably used as the terminal electron acceptor in aerobic bacteria and facultative anaerobes, because it generates the greatest Gibbs free energy alter and produces the most free energy.[2]
In anaerobic environments, different, lower-energy electron acceptors are used, including nitrate, nitrite, ferric iron, sulfate, carbon dioxide, and small organic molecules such as fumarate.
Photosynthetic [edit]
In oxidative phosphorylation, electrons are transferred from a moderate-energy electron donor such as NADH to a high-energy acceptor such equally O2 through an electron transport chain, releasing the energy. In photophosphorylation, the free energy of sunlight is used to create a high-free energy electron donor which can later on reduce oxidized components and couple to ATP synthesis via proton translocation by the electron transport chain.[10]
Photosynthetic electron ship chains, like the mitochondrial chain, can exist considered as a special instance of the bacterial systems. They employ mobile, lipid-soluble quinone carriers (phylloquinone and plastoquinone) and mobile, water-soluble carriers (cytochromes). They as well incorporate a proton pump. The proton pump in all photosynthetic chains resembles mitochondrial Complex III. The ordinarily-held theory of symbiogenesis proposes that both organelles descended from bacteria.
See also [edit]
- Charge-transfer complex
- CoRR hypothesis
- Electron equivalent
- Hydrogen hypothesis
- Respirasome
References [edit]
- ^ Lyall, Fiona (2010). "Biochemistry". Basic Science in Obstetrics and Gynaecology. pp. 143–171. doi:x.1016/B978-0-443-10281-3.00013-0. ISBN978-0-443-10281-3.
- ^ a b c Schmidt-Rohr Yard (Feb 2020). "Oxygen Is the Loftier-Energy Molecule Powering Circuitous Multicellular Life: Primal Corrections to Traditional Bioenergetics". ACS Omega. v (5): 2221–2233. doi:10.1021/acsomega.9b03352. PMC7016920. PMID 32064383.
- ^ a b Anraku Y (June 1988). "Bacterial electron ship chains". Almanac Review of Biochemistry. 57 (1): 101–32. doi:10.1146/annurev.bi.57.070188.000533. PMID 3052268.
- ^ Kracke F, Vassilev I, Krömer JO (2015). "Microbial electron send and energy conservation - the foundation for optimizing bioelectrochemical systems". Frontiers in Microbiology. 6: 575. doi:ten.3389/fmicb.2015.00575. PMC4463002. PMID 26124754.
- ^ Waldenström JG (2009-04-24). "Biochemistry. By Lubert Stryer". Acta Medica Scandinavica. 198 (i–vi): 436. doi:x.1111/j.0954-6820.1975.tb19571.ten. ISSN 0001-6101.
- ^ Zorova LD, Popkov VA, Plotnikov EY, Silachev DN, Pevzner IB, Jankauskas SS, et al. (July 2018). "Mitochondrial membrane potential". Analytical Biochemistry. 552: l–59. doi:10.1016/j.ab.2017.07.009. PMC5792320. PMID 28711444.
- ^ Lauren, Biochemistry, Johnson/Cole, 2010, pp 598-611
- ^ Garrett & Grisham, Biochemistry, Brooks/Cole, 2010, pp 598-611
- ^ Garrett R, Grisham CM (2016). biochemistry. Boston: Cengage. p. 687. ISBN978-i-305-57720-6.
- ^ a b Stryer. Biochemistry. toppan. OCLC 785100491.
- ^ Jonckheere AI, Smeitink JA, Rodenburg RJ (March 2012). "Mitochondrial ATP synthase: architecture, part and pathology". Journal of Inherited Metabolic Disease. 35 (2): 211–25. doi:x.1007/s10545-011-9382-ix. PMC3278611. PMID 21874297.
- ^ a b Garrett RH, Grisham CM (2012). Biochemistry (fifth ed.). Cengage learning. p. 664. ISBN978-1-133-10629-6.
- ^ Fillingame RH, Angevine CM, Dmitriev OY (November 2003). "Mechanics of coupling proton movements to c-ring rotation in ATP synthase". FEBS Letters. 555 (1): 29–34. doi:10.1016/S0014-5793(03)01101-3. PMID 14630314. S2CID 38896804.
- ^ Berg JM, Tymoczko JL, Stryer L (2002-01-01). "A Proton Gradient Powers the Synthesis of ATP".
- ^ Cannon B, Nedergaard J (January 2004). "Dark-brown adipose tissue: part and physiological significance". Physiological Reviews. 84 (i): 277–359. doi:10.1152/physrev.00015.2003. PMID 14715917.
- ^ Kim BH, Gadd GM (2008). "Introduction to bacterial physiology and metabolism". Bacterial Physiology and Metabolism. Cambridge Academy Printing. pp. ane–6. doi:ten.1017/cbo9780511790461.002. ISBN978-0-511-79046-i.
- ^ Mills EL, Kelly B, Logan A, Costa As, Varma M, Bryant CE, et al. (October 2016). "Succinate Dehydrogenase Supports Metabolic Repurposing of Mitochondria to Drive Inflammatory Macrophages". Cell. 167 (ii): 457–470.e13. doi:10.1016/j.cell.2016.08.064. PMC5863951. PMID 27667687.
- ^ EC i.3.v.ane
- ^ Ingledew WJ, Poole RK (September 1984). "The respiratory chains of Escherichia coli". Microbiological Reviews. 48 (3): 222–71. doi:10.1128/mmbr.48.iii.222-271.1984. PMC373010. PMID 6387427.
Further reading [edit]
- Fenchel T, Rex GM, Blackburn Th (September 2006). Bacterial Biogeochemistry: The Ecophysiology of Mineral Cycling (2nd ed.). Elsevier. ISBN978-0-12-103455-ix.
- Lengeler JW (January 1999). Drews G; Schlegel HG (eds.). Biology of the Prokaryotes. Blackwell Science. ISBN978-0-632-05357-5.
- Nelson DL, Cox MM (Apr 2005). Lehninger Principles of Biochemistry (quaternary ed.). Westward. H. Freeman. ISBN978-0-7167-4339-2.
- Nicholls DG, Ferguson SJ (July 2002). Bioenergetics 3. Academic Press. ISBN978-0-12-518121-1.
- Stumm W; Morgan JJ (1996). Aquatic Chemistry (3rd ed.). John Wiley & Sons. ISBN978-0-471-51185-four.
- Thauer RK, Jungermann Thou, Decker One thousand (March 1977). "Free energy conservation in chemotrophic anaerobic bacteria". Bacteriological Reviews. 41 (i): 100–80. doi:10.1128/MMBR.41.1.100-180.1977. PMC413997. PMID 860983.
- White D (September 1999). The Physiology and Biochemistry of Prokaryotes (2nd ed.). Oxford Academy Press. ISBN978-0-19-512579-5.
- Voet D, Voet JG (March 2004). Biochemistry. Biochemical Education. Vol. 28 (3rd ed.). John Wiley & Sons. pp. 124. doi:10.1016/s0307-4412(00)00032-vii. ISBN978-0-471-58651-7. PMID 10878303.
- Kim HS, Patel K, Muldoon-Jacobs K, Bisht KS, Aykin-Burns N, Pennington JD, et al. (Jan 2010). "SIRT3 is a mitochondria-localized tumor suppressor required for maintenance of mitochondrial integrity and metabolism during stress". Cancer Cell. 17 (1): 41–52. doi:ten.1016/j.ccr.2009.11.023. PMC3711519. PMID 20129246.
- Raimondi V, Ciccarese F, Ciminale V (January 2020). "Oncogenic pathways and the electron transport concatenation: a dangeROS liaison". Br J Cancer. 122 (ii): 168–181. doi:x.1038/s41416-019-0651-y. PMC7052168. PMID 31819197.
External links [edit]
- Electron+Transport+Concatenation+Complex+Proteins at the US National Library of Medicine Medical Subject Headings (MeSH)
- Khan Academy, video lecture
rosenbergmoodderge.blogspot.com
Source: https://en.wikipedia.org/wiki/Electron_transport_chain








0 Response to "what happens to the electrons as they move down the electron transport chain"
Publicar un comentario